Materials to capture CO2 from flue gas can be made to operate at up to 50 degrees lower temperatures by adding small amounts of transition metals such as zinc or nickel, report DIFFER researchers Mike Gleeson, Tesfaye Belete and Richard van de Sanden in the Journal of CO2 Utilization.
The finding can lead to more energy efficient carbon capture from flue gas, as well as to longer lived capture materials by reducing degradation from operating at high temperatures. In follow-up work, DIFFER will investigate how to convert the captured CO2 into high value chemicals or even clean fuels during the release from the carbon capturing material.

Making carbon capture more efficient and robust
The research by Mike Gleeson, Tesfaye Belete and Richard van de Sanden suggests a way to improve existing materials that capture CO2 from concentrated, industrial exhaust. The team managed to lower the temperature at which the materials release CO2 by up to 50 degrees from the regular 900-950 oC by adding small doses of transition metals (zinc, iron, cobalt, nickel or copper) to readily available carbon capture materials.
The finding not only reduces the amount of energy required for carbon capture. A drawback of carbon capture materials is that they degrade over multiple catch-and-release cycles because the high temperatures sinter the material so that it loses its open structure. Lowered operating temperatures thus open the way to longer lived materials.
Physicist Mike Gleeson: "The higher the temperature, the faster the degradation, so operating at a lower temperature would definitely make the material last longer. In follow-up work, we will put this claim to the test. Sneak preview: our preliminary work shows that zinc in particular has a dramatic effect that persists during repeated cycling."

Future work: clean fuels from captured CO2
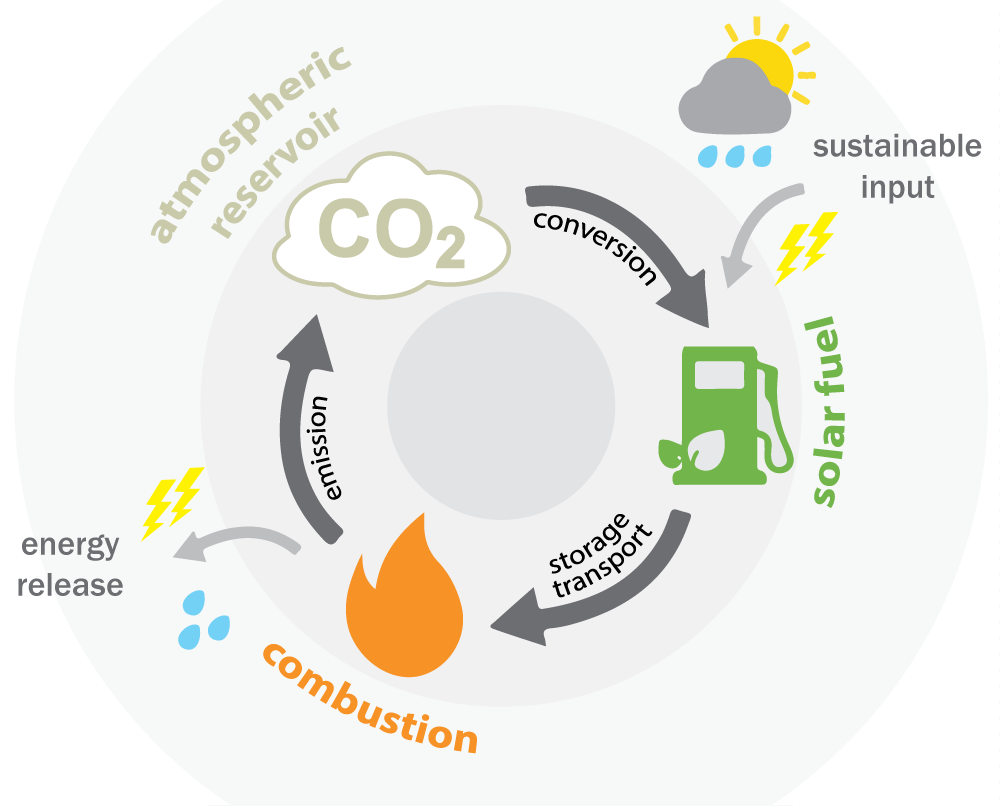
While increasing the efficiency of carbon capture is valuable in its own right, DIFFER's solar fuels research aims for a more ambitious goal: using captured CO2 together with renewable electrical power into fuels or other valuable chemicals.
"Solar fuels pack over forty times the amount of energy as the same weight of batteries", says Richard van de Sanden, who heads DIFFER's solar fuels research: "They offer a way to bridge seasonal gaps in energy storage, but also to power long range transport such as intercontinental flights, and weaning industry off of fossil feedstocks".
In their publication, the researchers showed that releasing CO2 from zinc-doped capture materials into a hydrogen atmosphere converts the greenhouse gas into carbon monoxide (CO). That is a crucial feedstock molecule for the production of many fuels such as methane, diesel or kerosene, but also plastics and medicine. Further research will look into providing the necessary hydrogen by performing the reaction in a plasma (ionized gas) of water molecules.
Richard van de Sanden: "CO2 is too valuable to just pump into the atmosphere - or store it underground, for that matter. We need to enable a circular carbon infrastructure where any emitted CO2 is constantly recycled into new fuels and chemicals for our economy. That is the only way to truly stop using fossil resources."
About DIFFER
The Dutch Institute for Fundamental Energy Research DIFFER is one of the nine research institutes of the Netherlands Organisation for Scientific Research NWO. DIFFER performs cross-disciplinary research on materials, processes and systems for a global sustainable energy infrastructure, in close partnership with (inter)national academia and industry. The research at DIFFER covers both the conversion and storage of sustainable energy in solar fuels, and the generation of clean, safe and abundant power through nuclear fusion.
Go to the News page.